Lab of Brain Evo-devo
Decoding brain evo-devo
The human brain has undergone profound multi-dimensional remodeling throughout evolution, characterized by expanded brain volume, increased cortical gyrification, enhanced complexity in neuronal morphology and connectivity, and significantly protracted development. While these features form the structural and developmental foundation for the emergence of higher cognition, this intricate complexity comes with an evolutionary cost: increased susceptibility to cognitive disorders such as ASD, Schizophrenia, and Alzheimer’s Disease (see our review in Curr. Opin. Neurobiol., 2023). Adopting an evolutionary-developmental (evo-devo) perspective allows us to elucidate how the structural basis of human cognition is constructed during development and how perturbations in these programs translate into disease risk. To address these core scientific questions, our laboratory has established a comprehensive multi-species research platform covering mice, pigs, marmosets, macaques, and humans. By integrating multi-scale data—spanning genomics, epigenetics, transcriptomics, proteomics, and metabolomics—with deep learning and AI models, we systematically investigate the mechanisms underlying human brain uniqueness and the evolution of susceptibility to brain disorders.
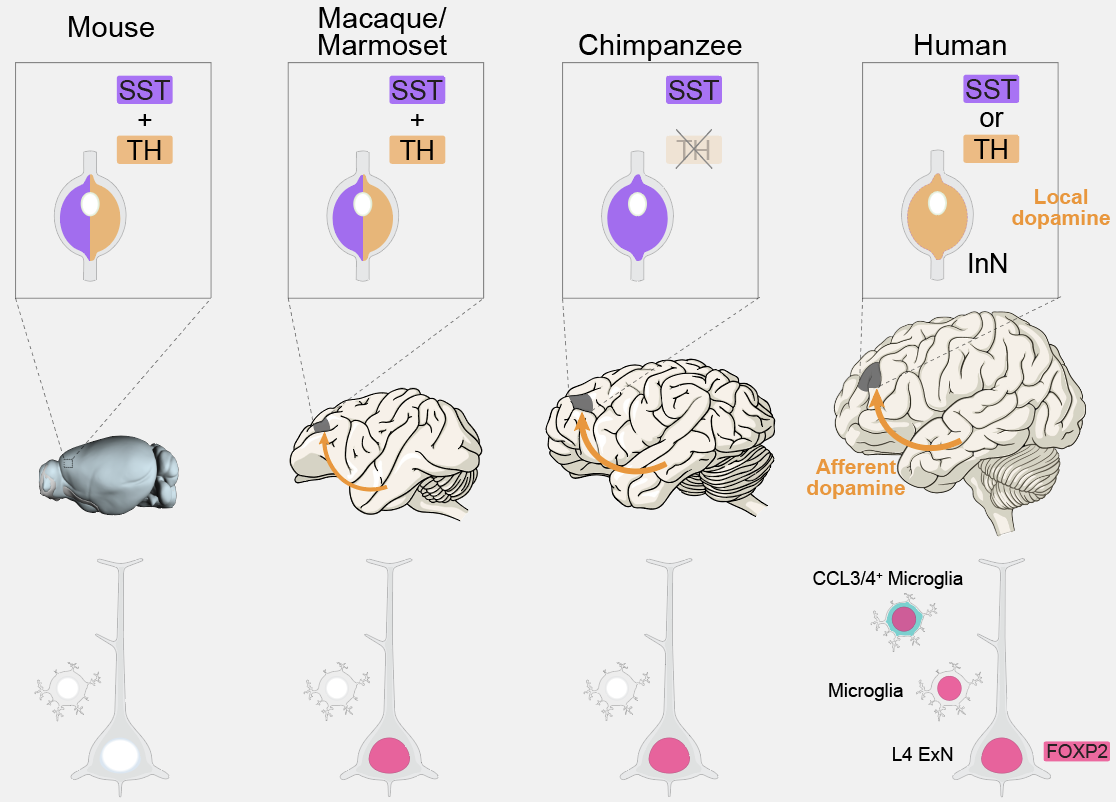
Evo-devo Mechanisms Underlying Uniqueness of Human Brain
Focused on “What makes us human,” we systematically profile human-specific characteristics and dissect their molecular and cellular mechanisms through cross-species and cross-development comparisons. Previously, we identified multi-faceted human-specific features—including distinct cell types, dopamine-secreting interneurons, and the expression regulation of language-related genes—and discovered the GALP signaling pathway as a key driver of prefrontal cortex expansion (Science 2022, 2023). Current research aims to address remaining unknowns, such as the regulatory networks driving protracted brain maturation, how this prolonged window impacts plasticity and functional networks, and the precise control of the assembly of human-specific neural circuits. We are combining comparative genomics with spatiotemporal multimodal profiling to understand how genomic variants and regulatory remodeling synergistically influence neuronal maturation and circuit assembly, tracing the evolutionary origins of higher-order cognitive functions from molecules to circuits.
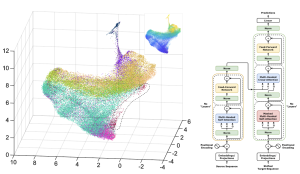
In silico Modeling of Human Brain Evo-Devo
The formation of the human brain’s unique structure and developmental patterns stems from systemic remodeling of gene regulatory networks rather than single-point mutations. Similarly, cognitive disorders involve complex mechanisms coupling multiple genes, pathways, and scales. To deeply understand these regulatory laws and their causal links to disease susceptibility, high-dimensional integration of multi-scale information is essential. Leveraging the rapid accumulation of cross-species multi-omics data, we aim to build and integrate resources spanning the “molecule-cell-connectivity” scales. By fusing multi-modal data with deep learning algorithms and Large Language Model frameworks, we are developing digital models of human brain evolution and development. This approach enables the systematic screening and interpretable prediction of key developmental regulators and potential pathogenic elements, which are then validated experimentally to form a closed-loop discovery process.
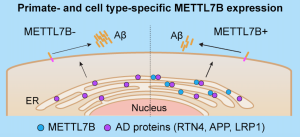
Evolutionary Medicine for Brain Disorders
Evolution has shaped natural heterogeneity in disease susceptibility across populations and species. This heterogeneity is not noise but a vital clue for understanding disease mechanisms and guiding interventions. For instance, we discovered that the primate-specific methyltransferase METTL7B confers neuroprotection, likely representing an adaptive response to the increased vulnerability to aging and AD in primates (Neuron 2022). Our laboratory is currently utilizing our multi-species platform to systematically analyze the key cell types and molecular pathways driving evolutionary differences in disease susceptibility. Based on these insights, we aim to develop novel, translatable therapeutic strategies and intervention targets for aging and cognitive disorders.